By Dr. Marco V. Benavides Sánchez.
Lung transplantation remains the only definitive therapy for many patients with end-stage lung disease. However, the shortage of suitable human donor lungs continues to be one of the most pressing issues in modern medicine. Thousands of patients die each year while waiting for a transplant, and even those who receive a human lung often face significant complications, including graft dysfunction and chronic rejection. Against this backdrop, xenotransplantation—the transplantation of organs from animals into humans—has emerged as a potential solution.
Recent progress in genetic engineering of pigs has revived interest in this field, as pigs are physiologically compatible in size and function with humans, and their genome can be modified to reduce immune incompatibility. On August 25, 2025, a groundbreaking report in Nature Medicine described the first-ever case of pig-to-human lung xenotransplantation into a brain-dead human recipient. The work, led by Jianxing He and an international team of collaborators, represents a historic step forward, though many challenges remain before clinical application becomes a reality.
This article provides a detailed review of the study, its methodology, findings, and implications for the future of lung xenotransplantation.
The Study Design
The case involved transplanting a lung from a six-gene-edited pig into a 39-year-old brain-dead male recipient who had suffered a fatal brain hemorrhage. Unlike previous xenotransplantation studies that focused on kidneys or hearts, this was the first time a genetically engineered pig lung was tested in a human.
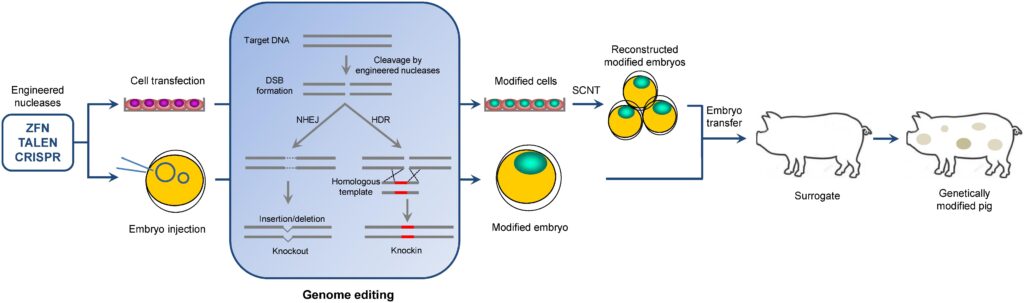
The monitoring period lasted 216 hours (9 days), during which the lung’s viability and functionality were carefully assessed. Researchers were particularly interested in observing whether the human immune system would launch an immediate, catastrophic response—known as hyperacute rejection—which has historically been the major barrier to xenotransplantation.
Immunosuppressive Protocol
To prevent rejection, the team employed an aggressive multi-agent immunosuppressive regimen, including:
- Rabbit anti-thymocyte globulin – for T-cell depletion
- Basiliximab – an interleukin-2 receptor antagonist
- Rituximab – targeting B cells
- Eculizumab – a complement inhibitor
- Tofacitinib – a JAK inhibitor
- Tacrolimus – a calcineurin inhibitor
- Mycophenolate mofetil – an antiproliferative agent
- Steroids – tapered over time
Adjustments to the regimen were made based on ongoing assessments of the recipient’s immune status. This complex cocktail reflects the current uncertainty about how the human immune system will respond to a xenograft and highlights the need for further refinement.
Key Observations
1. Absence of Hyperacute Rejection
One of the most encouraging findings was that the transplanted lung did not undergo hyperacute rejection. This suggests that the six genetic modifications made to the pig donor were effective in minimizing the most immediate and devastating immune reactions.
2. Severe Edema at 24 Hours
By 24 hours post-transplantation, the lung developed severe edema resembling primary graft dysfunction, a complication commonly seen in human lung transplants. The researchers hypothesized that this was due to ischemia–reperfusion injury, a process that damages tissue when blood supply returns after a period of deprivation.
3. Antibody-Mediated Rejection (AMR)
By postoperative days 3 and 6, there was evidence of antibody-mediated rejection, which caused tissue damage in the xenograft. Interestingly, partial recovery was observed by day 9, suggesting that rejection was not uniformly progressive and could be modulated by immunosuppression.
4. Infection Surveillance
Importantly, there were no signs of zoonotic infection during the observation period. However, the long-term risk of porcine endogenous retroviruses (PERVs) and other cross-species pathogens remains a major concern for clinical transplantation.
Significance of the Findings
This study provides proof of concept that genetically engineered pig lungs can function in a human thoracic cavity, at least temporarily, without catastrophic immune rejection. While the xenograft eventually suffered immune-mediated damage, the fact that it survived and functioned for nine days marks a historic advance.
It also provides researchers with valuable insights into the unique immunological dynamics of lung xenotransplantation, which appear to differ significantly from kidney or heart xenografts. For instance, the lungs are constantly exposed to external air, making them particularly vulnerable to immune activation and infection.
Challenges Ahead
Despite the success, several formidable barriers must be overcome before pig-to-human lung transplantation becomes a clinical option:
1. Immune Compatibility
While genetic engineering has reduced hyperacute rejection, other forms of rejection—particularly antibody-mediated and chronic rejection—remain significant risks. More precise genetic editing may be needed to further humanize pig organs.
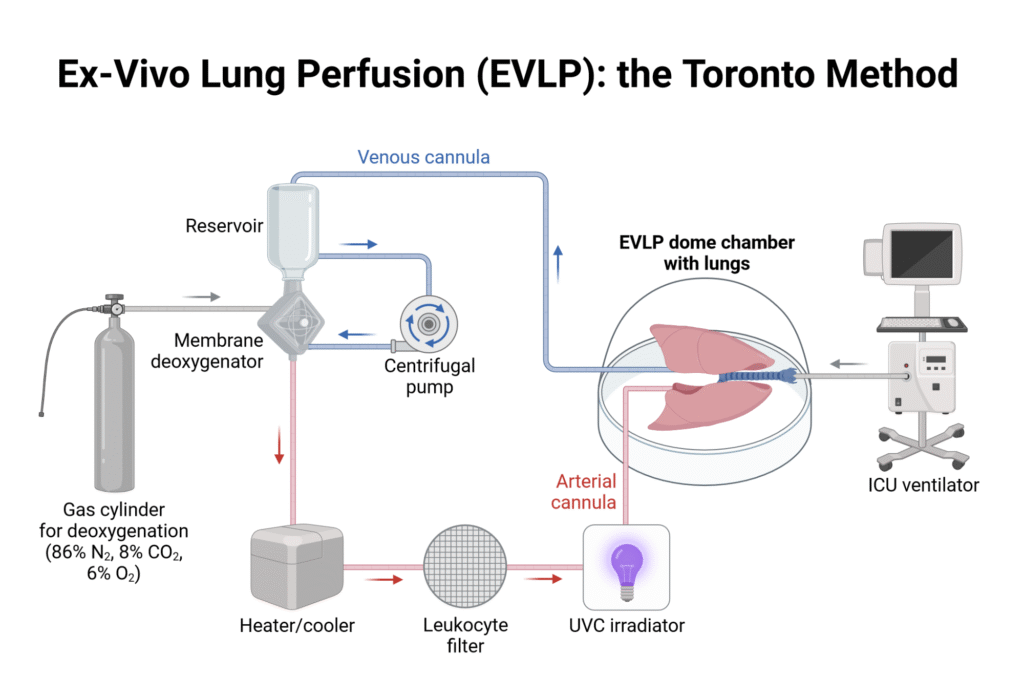
2. Ischemia–Reperfusion Injury
Strategies to minimize this type of injury will be crucial. Techniques such as ex vivo lung perfusion (EVLP) may help condition xenografts before transplantation.
3. Infection Risk
Preventing zoonotic infections is essential for both recipient safety and public health. Ongoing efforts to inactivate porcine endogenous retroviruses using CRISPR-Cas9 may mitigate this risk.
4. Immunosuppressive Toxicity
The regimen used in this case was intensive and would likely be unsustainable in a living patient. Future protocols must find a balance between efficacy and safety.
5. Ethical and Regulatory Issues
Xenotransplantation raises profound ethical questions regarding animal welfare, patient consent, and societal risk. Rigorous oversight and public dialogue will be needed as the field advances.
Broader Context in Xenotransplantation
This landmark study follows other recent xenotransplant milestones. In 2022 and 2023, pig kidneys and hearts were successfully transplanted into brain-dead and living human recipients, sparking international attention. The lung, however, presents unique challenges because of its complexity, fragility, and exposure to the environment.
Thus, while pig heart and kidney xenotransplantation may reach clinical use sooner, the feasibility demonstrated in this study ensures that lung xenotransplantation will remain an active area of research.
Future Directions
Several strategies are likely to accelerate progress:
- Advanced Gene Editing: Incorporating more extensive edits to reduce antigenicity and improve compatibility.
- Tolerance Induction: Exploring ways to retrain the human immune system to accept xenografts, potentially using cell therapy or regulatory T cells.
- Bioengineered Scaffolds: Combining pig-derived scaffolds with human stem cells to create hybrid organs.
- Longer Observation Studies: Future research should extend monitoring beyond 9 days to assess longer-term viability.
Conclusion
The report by He et al. (2025) marks a watershed moment in transplant medicine. For the first time, a genetically engineered pig lung was transplanted into a human recipient, demonstrating short-term viability and resistance to hyperacute rejection. While significant obstacles remain—including antibody-mediated rejection, infection risk, and the need for safer immunosuppression—this study represents a critical proof of concept.
The path to clinical lung xenotransplantation will likely be long and complex, but this breakthrough underscores the immense potential of xenotransplantation to one day alleviate the global organ shortage and save countless lives.
Reference
- He, J., Shi, J., Yang, C., Peng, G., Ju, C., Zhao, Y., Liu, H., He, P., Liu, X., Zhang, Z., Chen, C., Pan, D., Yang, Z., Guang, W., Li, H., Chen, Z., Liu, M., Liang, H., Huang, W., Jeon, K., Chen-Yoshikawa, T. F., Rucker, A. J., Lal, A., Zhong, N., Zhang, K., Liu, X., & Xu, X. (2025). Pig-to-human lung xenotransplantation into a brain-dead recipient. Nature Medicine. https://doi.org/10.1038/s41591-025-03861-x
#ArtificialIntelligence #Medicine #Surgery #Medmultilingua


Leave a Reply